Product and Market Opportunities
The TRS technology platform is ideally suited for a variety of important clinical applications. Our long term objective is to partner with major medical device, dental and pharmaceutical companies to develop comprehensive skeletal reconstruction product lines targeted at five distinct markets, including craniomaxillofacial (CMF) surgery, orthopedic surgery, spine fusion surgery, dental implant surgery, and bone growth factor delivery.
CMF Surgery
This has been a primary focus of our research and product development efforts over the last several years. For complex CMF skeletal repair or reconstruction, the current surgical gold standard is the implantation of autologous bone graft, harvested from the patient's hip, fibula or scapula, and anchored by metal plate and screw systems. Other indications, such as cranioplasty, are addressed by the use of metal or polymer plates or meshes.
Implants constructed from the TRS scaffold technology offer compelling advantages for CMF surgery. First, they have significant structural integrity to bear load and support function, but are bioresorbable and fully replaced over time with natural bone. Second, they eliminate the need for autologous bone graft harvest, a secondary surgery that is time consuming, costly, and frequently associated with complications. Third, their geometry can be custom constructed to fit the patient’s defect, eliminating the need for time consuming intra-operative carpentry. Finally, they are differentiated by their Affinity™ coating that actively promotes natural bone formation, accelerating healing and reducing the need to use expensive bone growth factor products.
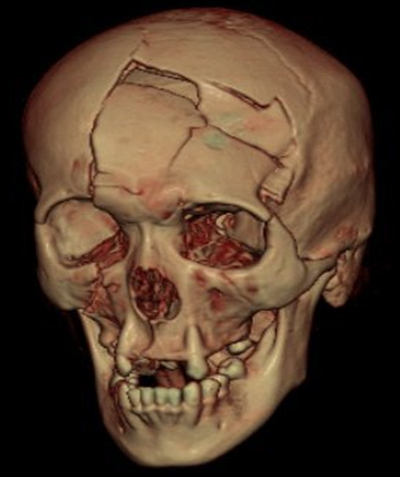 |
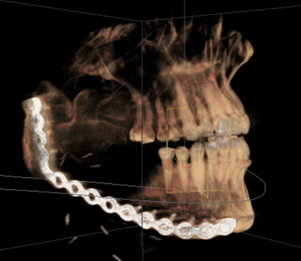 |
| Complex Cranial Bone Trauma | Excised Mandible Tumor |
Orthopedic Surgery
Similar to CMF surgery, the TRS scaffold technology is an ideal platform for the production of patent specific implants to treat complex skeletal defects resulting from trauma or tumor removal.
Soft tissue injury repair is another orthopedic surgery procedure where TRS technology can advance patient care. In this procedure, the clinical challenge is to rapidly restore a strong attachment of soft tissue structures to bone, a common requirement in rotator cuff surgery as well as reconstructive surgery for knee ligament repair. The application of our Affinity™ coating to conventional bone anchors will accelerate bone growth and healing. Further, in vivo studies have shown that sutures coated with Affinity™ can accelerate early bone-tendon and tendon-tendon healing compared to uncoated sutures. In addition, Affinity™ provides a mechanism to bind autologous biologics and extend their effects for longer timeframes, thereby providing a biologic stimulus to healing.
The TRS Affinity™ coating can also be applied to the fixation surfaces of metallic hip and knee replacement implant systems to accelerate bone formation and strengthen permanent attachment.
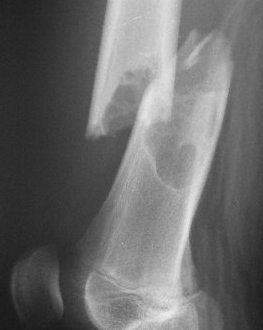 |
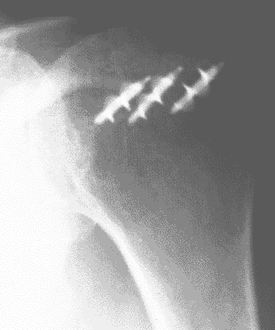 |
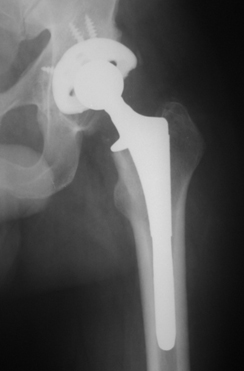 |
| Long Bone Tumor Resulting in Fracture |
Rotator Cuff Re-Attachment to Bone |
Hip Replacement Implant Anchored in Femur |
Spine Fusion Surgery
Spine fusion is the gold standard therapy for serious degenerative conditions of the cervical and lumbar spine. There are a variety of implant technologies and products currently marketed for insertion into the disc space as part of the spine fusion procedure. These include metallic cages, spacers made of PEEK, an inert, plastic material, and dowels constructed from cadaveric bone. While fusion implant technologies may differ, their common purpose is to stabilize the disc space and maintain proper spacing between vertebral bodies while bone growth occurs to form a solid bony bridge that fuses the two vertebra together. In addition to the interbody space, spine surgeons often place bone graft or bone substitute materials in the adjacent "lateral gutter" regions to enhance and widen the fusing bone bridge.
The TRS scaffold technology can be used to construct cervical interbody fusion cages that have important clinical advantages. First, unlike metallic or PEEK cages, they are bioresorbable over time, so no foreign material remains in the disc space once the fusion process is complete. Second, they are differentiated by their Affinity™ coating and its ability to bind and release bone growth factors in a more controlled fashion, lessening problematic side effects such as heterotopic bone formation and systemic dispersion of the growth factor. Finally, they closely resemble cadaveric bone dowels in both form and function, but are synthetic and do not have the sourcing, variations in quality, processing challenges, or expense associated with the use of cadaveric material. Our scaffold technology can be also be used to construct novel lateral gutter implants with the same attributes.
Our Affinity™ coating can be grown on the surfaces of conventional interbody fusion implants constructed of Titanium or PEEK, as well as currently marketed synthetic bone graft materials. Adding our coating to these products will give them enhanced bioactive properties, including the ability to be intraoperatively loaded with a growth factor and deliver it locally, in a controlled release fashion.

TRS Cervical Interbody Fusion Cage and Lateral Gutter Implant |

Titanium and PEEK Interbody Fusion Cages |
Dental Implant Surgery
In complex cases involving significant underlying bone deterioration, dental implant surgery is usually performed in two stages. The objective of the first stage procedure is to restore proper bone volume and geometry to support the dental implants. This is accomplished by either implanting bone blocks sourced from the patient, or some combination of bone graft and synthetic bone substitute material in conjunction with metal scaffold meshes. Once bone growth and stability are achieved, usually over a six to nine-month period, the second stage procedure to remove metal hardware and install the dental implants is performed. TRS scaffold technology can be used to construct patient specific implants for dental bone restoration with distinct advantages. First, they eliminate the need to harvest autologous bone blocks and the associated complications. Second, unlike metal mesh implants, they are bioresorbable and do not need to be removed. Third, they are custom designed from patient imaging studies. allowing for an exact fit and optimal geometry. Finally, they are differentiated by their Affinity™ coating that actively promotes natural bone formation, accelerating the timetable for the second stage procedure.
In addition to TRS scaffold opportunities, our Affinity™ coating can be grown on the fixation surfaces of dental implants to give them the same enhanced bioactive properties as TRS bioresorbable implants.
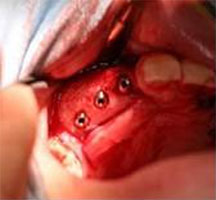
Autologous Bone Block |

Dental Bone Restoration Implant |
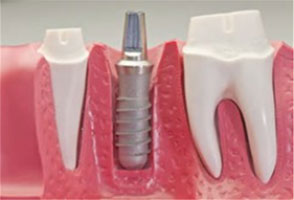
Dental Implant Fixation Post |
Bone Growth Factor Delivery
Bone growth factors offer important clinical advantages and patient care benefits. However, they are also associated with problematic side effects. Most of these side effects stem from the use of direct application or collagen sponges to deliver the growth factor. These delivery techniques result in dosage variances and bolus release which can lead to unwanted dissipation of the growth factor outside of the desired delivery location. To make growth factors safer, there is an urgent need for improved delivery techniques that eliminate bolus release, lower dosage requirements, contain delivery to the target area, and prevent systemic dissipation. Another limitation of current growth factor delivery strategies, like the use of collagen sponges, is the lack of structure. Structural implants that carry and deliver growth factors have the potential to significantly expand the opportunities for their use as well as optimize outcomes.
Our research has clearly demonstrated that coated TRS scaffolds serve as an excellent substrate for adhesion and local, controlled release of bone growth factors. TRS technology can be used to construct structural carriers uniquely and specifically suited for a wide variety of clinical applications. TRS also has research experience with, and patents covering "microsphere" technology that binds growth factors and delivers them via injection with the same controlled release advantages. In summary, TRS technology represents a significantly improved and safer delivery vehicle for bone growth factors that can expand their indications and market adoption.

