Foundational Research
The TRS technology platform has been the subject of comprehensive academic, NIH and company-sponsored research, spanning a period of well over a decade. Research initiatives have included technology-specific studies, basic bench testing of fabricated implants and their coating science, studies validating the application of our Affinity™ coating to other bioresorbable materials as well as Titanium and PEEK implants, and large animal implant studies with long-term follow-up. Several publications have resulted from this body of research that clearly demonstrate the unique attributes of our technology platform and its clinical viability.
Bone Tissue Engineering Using Polycaprolactone Scaffolds Fabricated Via Selective Laser Sintering
Biomaterials 26 (2005) 4817-4827
This early research completed by the TRS founders at the University of Michigan focused on implant design and fabrication, micro-architecture and mechanical property assessment, and demonstration of implant-stimulated bone growth. Published in 2005, this research illustrated the ability to design and fabricate bioresorbable implants with porous micro-architectures possessing sufficient mechanical properties for bone tissue engineering applications. Further, histological evaluation and CT scan analysis showed that bone growth can be generated in vivo.
Computed Tomography-Based Tissue Engineered Scaffolds in Craniomaxillofacial Surgery
The International Journal of Medical Robotics and Computer Assisted Surgery 3 (2007) 207-216
In this study, published in 2007, mandibular condyle replacement implants were designed and fabricated directly from CT scans and implanted to reconstruct the temporomandibular joint in Yucatan Mini-Pigs. These implants precisely matched the pig mandibular anatomy and allowed for proper surgical fixation. Following sacrifice, CT scanning and histological evaluation were used to interrogate the efficacy of the implant. The study illustrated the ability to design and fabricate temporomandibular joint replacement implants that were able to support masticatory function in a large animal model. Further, cartilaginous tissue regeneration along the articulating surface, and exuberant bone tissue formation were noted.
 |
|
TRS Cervical Fusion Cage Implant |
A new Resorbable Integrated Cervical Plate/Cage Fusion Device Modified with Osteoconductive Coating or Osteoinductive Factors: Preliminary Results in a Pre-Clinical Yucatan Minipig Model
In this study, which is pending publication, an anterior cervical fusion cage design was created using image-based methods and representative CT data from the Yucatan Mini-Pig. The cage was designed to withstand surgical impaction forces and to have strength characteristics within the range of vertebral body bone. A group of implants was mechanically tested in compression to verify achievement of this design goal. Cages were then either mineral coated or filled with a collagen sponge soaked in BMP-7, and implanted via an anterior cervical discectomy and fusion procedure performed on Yucatan Mini-Pigs at the C5-C6 level. Prior to sacrifice, CT scans were completed on anesthetized animals to assess bone in-growth. After sacrifice at 6, 12, or 18 months, the fusions were evaluated by CT scanning and the residual PCL material from each fusion was assessed to determine the degree of biodegradation. This research demonstrated that the TRS technology platform can be used to create anterior cervical fusion cages with strength attributes applicable for human use that facilitated successful fusion results when implanted in the pig model. Further, the addition of a mineral coating to the fusion cages was as effective as the addition of a growth factor in encouraging bone in-growth and fusion at later time points. Finally, degradation of the implant material was compatible with bone regeneration.
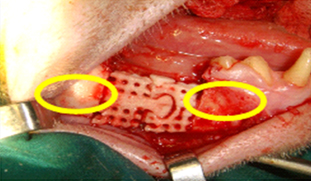 |
| Modular TRS Mandibular Implant with Press Fit Fixation Shown in Yellow. |
Mandibular Reconstruction in a Primate Model
In collaboration with the National Dental Center of Singapore, TRS designed and fabricated modular, osteoconductively coated implants that were used to reconstruct mandibular segmental defects in adult Macaca Fascicularis monkeys. In this study, 8 monkeys received scaffolds alone, 8 monkeys received scaffolds soaked intra-operatively with BMP2, and 8 monkeys received scaffolds coated with autologous bone marrow cells. This study is still ongoing, with longest follow-up now approaching two years. Results to date demonstrate the feasibility of successfully reconstructing mandibular segmental defects using modular TRS implants.
NIH Grant Studies
In the last two years TRS has been the recipient of large NIH research grants to evaluate both the biologic properties of our technology platform and our mandibular reconstruction implants.
TRS was awarded a $200,000 NIH Phase I grant to refine and evaluate intra-operative strategies to bind various bone growth factors to the surface of our CMF reconstruction implants. This research was successfully completed and all study endpoints were achieved.
As a follow-up to a prior and successful NIH Phase I project, TRS was awarded an $850,000 Phase II grant to clinically evaluate the efficacy of a modular, bioresorbable, osteoconductive TRS implant to reconstruct large mandibular defects in a Yorkshire pig model. Follow-up was taken out to one year. A new implant design was used in this study.
Affinity™ Research
TRS has refined the process to successfully apply our Affinity™ coating to Titanium implant surfaces. Further, we have conducted direct comparative studies of Affinity™ coated Titanium versus Titanium coated with Plasma Sprayed HA. These studies demonstrate comparable mechanical adhesion properties and show that cells attach to the Affinity™ coated surface more readily and rapidly than the Plasma Sprayed HA surface.
TRS has developed a proprietary method of producing our Affinity™ coating on PEEK that meets or exceeds ASTM coating adhesion tests. The TRS coating on PEEK transforms the implant surface from hydrophobic to hydrophilic, promoting osteoblastic differentiation and fostering a cellular environment that favors bone formation. In marked contrast to conventional PEEK, Affinity™ coated PEEK demonstrates 71-77% cell attachment within 45 minutes, with equally superior three and seven-day cell proliferation.
In the area of bone graft substitute materials, TRS has successfully applied the Affinity™ coating to a wide range of biomaterials, including beta-TCP, hydroxyapatite, PCL, PLGA, and alginate. Our proprietary methods enable us to conduct screening studies to optimize the short-term attachment and osteogenic differentiation of mesenchymal stem cells on TRS coated bone graft substitutes, rendering these materials osteostimulatory.
The Affinity™ coating technology was originally developed by TRS co-founder Bill Murphy, PhD at the University of Wisconsin Bioinspired Materials Laboratory. The extraordinary characteristics of this coating have been the subject of numerous published studies that can be viewed by visiting their website: http://bioinspired.engr.wisc.edu/.
![]() Read a published article by TRS founders discussing the field of bone tissue engineering.
Read a published article by TRS founders discussing the field of bone tissue engineering.

